 When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Only one ion of each is needed to balance these charges. If we look at the ionic compound consisting of lithium ions and bromide ions, we see that the lithium ion has a 1+ charge and the bromide ion has a 1− charge. This rule is ultimately based on the fact that matter is, overall, electrically neutral.īy convention, assume that there is only one atom if a subscript is not present. To determine the proper formula of any combination of ions, determine how many of each ion is needed to balance the total positive and negative charges in the compound. Finally, the proper formula for an ionic compound always obeys the following rule: the total positive charge must equal the total negative charge. 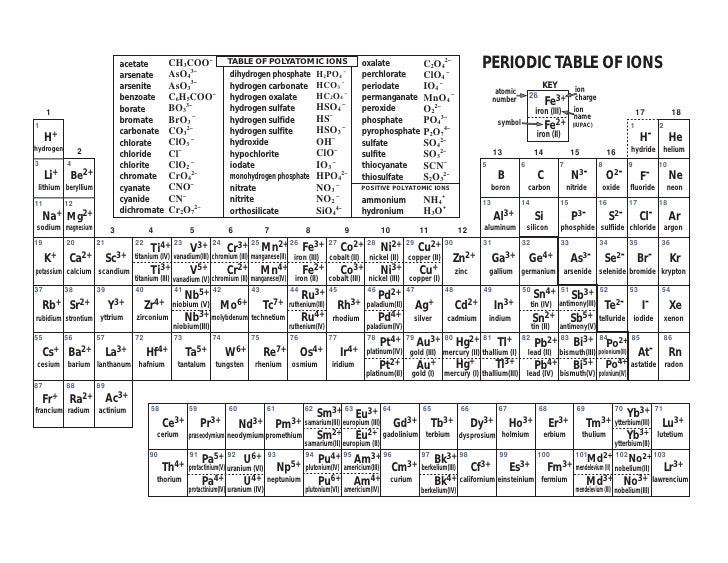
Remember that in an ionic compound, the component species are ions, not neutral atoms, even though the formula does not contain charges. Second, charges are not written in a formula. 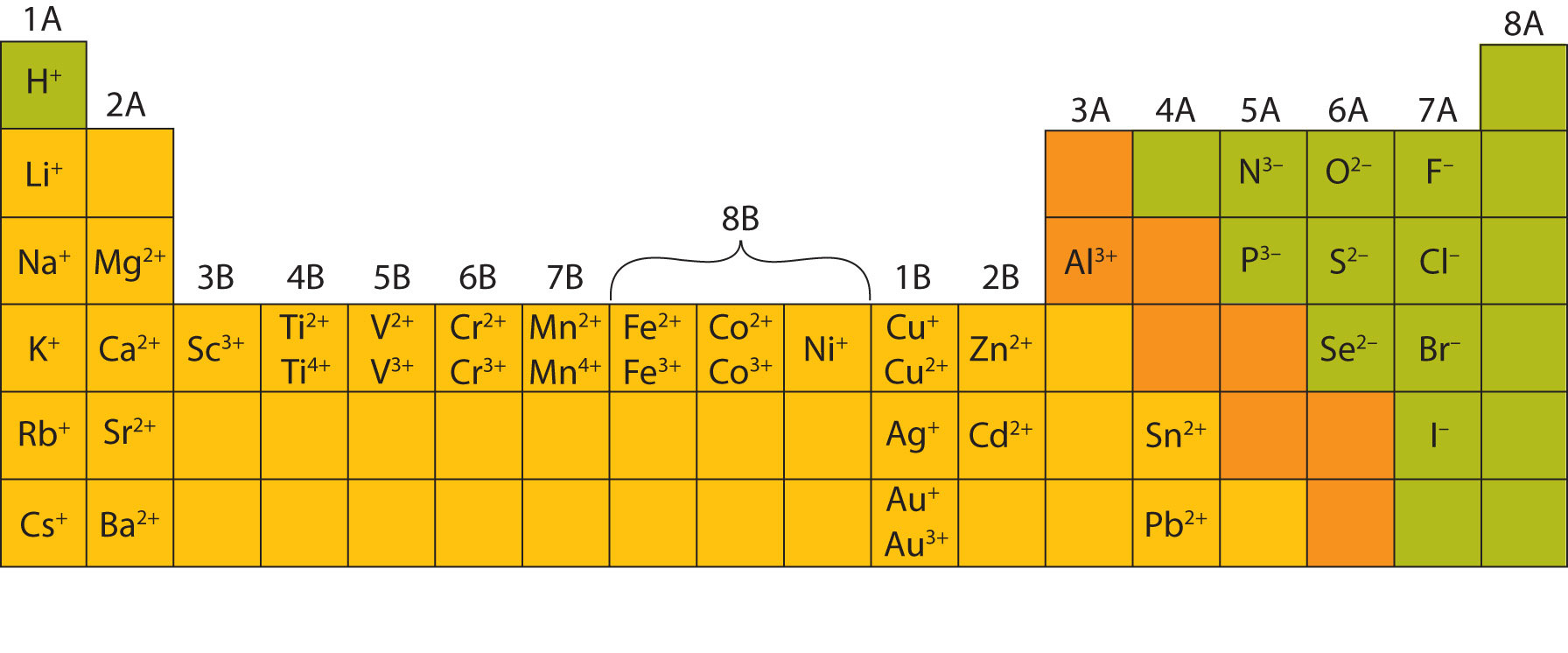
Because most metals form cations and most nonmetals form anions, formulas typically list the metal first and then the nonmetal. First, the cation is written before the anion. The formula for an ionic compound follows several conventions. 
A crystal of sodium chloride, shown here, is a collection of alternating sodium and chlorine ions. The precise pattern depends on the compound. A crystal contains a three-dimensional array of alternating positive and negative ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
